
medical devices and sterilization
Our goal is to provide a comprehensive offer for the client, taking into account safety and responsibility for the quality of products and services provided within the medical business.
We are the only company in Poland that offers comprehensive solutions, bearing in mind production of medical products, packing them in a safe medical packaging and sterilization using ethylene oxide or steam.

medical devices
Medical Business Unit Plastica Ltd. is the most modern factory in the European Union. Here is located the automated part of medical devices production, the remaining arise in our sister company in India. The production of medical devices places us under obligation to comply with precisely specified conditions of work. The production plant in Plastica Ltd. Medical Business Unit was designed to meet the requirements of Good Manufacturing Practice and the union PN-EN ISO 14644-1:2005 norm in class 8.

medical devices
The modern medical devices are produced in a clean room under the Matopat brand, with the application of modern machinery, since years they are used in surgical and treatment rooms, dispensaries, dialysis centers and in ambulances. Through long experience and cooperation with many authorities from medical field we prepared the wide range of products which are consistently developed.
The latest technical solutions implemented in Plastica Ltd. Medical Business Unit are efficient, fully automated, which helps to increase safety of medical devices and their availability to the customer.
The final stage of medical devices production is sterilization, which can be realized in both high and low temperatures, depending on the materials used for production. Plastica Ltd. has many years of experience in sterilization using ethylene oxide and steam, offering a sterilization service and comprehensive solution along with medical packaging produced in place under the brand BOM (Safe Medical Packaging).
ethylene oxide sterilization
Our company uses ethylene oxide to sterilize medical devices due to reasons such as: low production cost, very good penetration of the ethylene oxide, the possibility of sterilizing devices that undergo damage under the influence of high temperature, the lack of degradation under the influence of a sterilizing factor. Plastica Ltd. offers a gas sterilization service with the usage of ethylene oxide in accordance with the PN-EN ISO 11135-1:2009 norm. This is the first installation of low-temperature underpressure sterilization installation in Poland.
more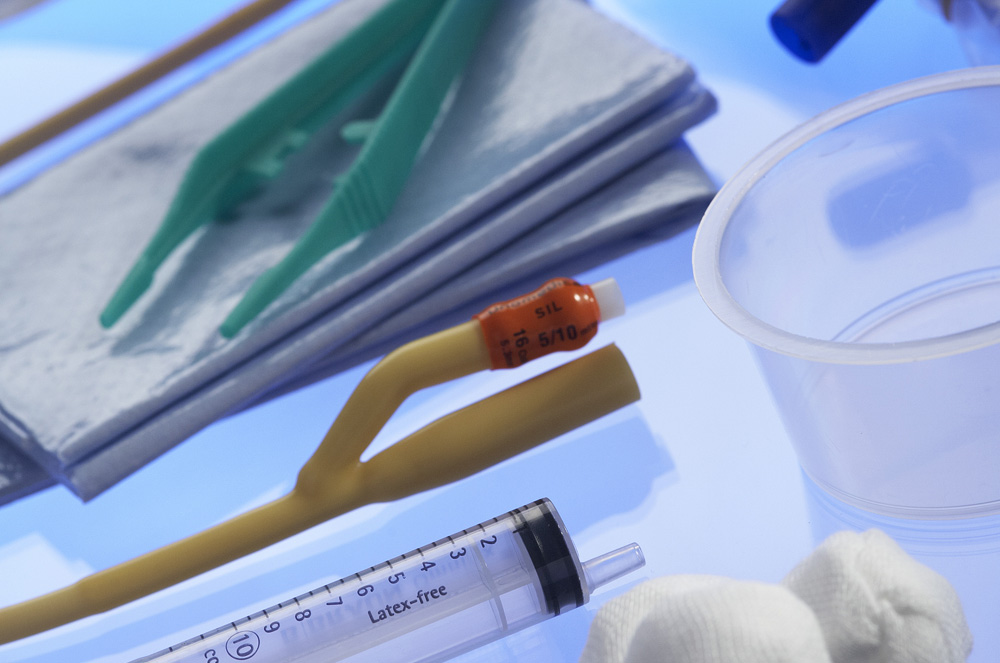

ethylene oxide sterilization
The sterilization process is preceded by the stage of initial conditioning, during which the micro-organisms undergo inducement. After the sterilization process is finished, the medical devices undergo quarantine period in the final conditioning room in order to degas the rest of the ethylene oxide. The whole process is monitored by a validated software.
New investment is fully automated process of sterilization in connection with initial conditioning and degassing process of ETO and ECH. Two chambers for ETO sterilization with capacity of 4 and 10 euro pallet give us enormous elasticity and competitive advantage in terms of logistics and costs of service.

steam sterilization
For products that do not require sterilization at low temperatures we offer sterilization using steam. Thermal sterilization in overpressure with usage of water vapour is the safest sterilization method of medical products and it’s universal and the most economical process.
more
steam sterilization
When choosing this method of sterilization it is very important to remember that the decisive factor in determining its effectiveness is high temperature therefore the product that is subjected to the process must be thermostable.
Another limitation is the fact that water vapour is a factor that works on a surface therefore such sterilization method is surface sterilization, which is used to sterilize both solid medical devices, as well as liquid and sealed in airtight containers.
With each new partner, our qualified specialists conduct the validation of sterilization process using common knowledge, experience and R-D and laboratory resources in the area of gas chromatography and microbiology, so that the process is performed correctly and effectively.
validation process
Sterilization process is subject to validation according to newest European and American requirements of the market. In order for the final product to be acknowledged as safe for final user and for the patient it is essential to proceed many tests and experiments. These activities are conducted by a validation team of engineers – practitioners. Long-term experience of our team in the area of chemistry, microbiology, process engineering, electronics and automation allow us to work out optimal parameters of products sterilization.


validation process
During design work and by cooperation with our clients it is determined the “worst case” which is the product with the biggest challenges and difficulties during sterilizing. After that the sterilization process (the set of required parameters) is selected on the basis of product’s structure, raw materials which it is built from and the way of packing. Qualified validation team of Plastica Ltd. is responsible for activities during the validation process such as: installation, operational and process qualification as well as all physical, chemical and microbiological tests. Our activities, long-term experience and professionalism have been appreciated by our clients and business partners.
products development inspired by practice
The priceless source of information on use of our medical devices in everyday medical practice is Matopat Hospital, belonging to the TZMO Holding Group. Hospital medical staff on a current basis gives us comments and guidance on the needs of medical institutions, and thus - suggestions for the further development of our products.
mocne strony
vitae mollis nibh
quisque at blandit justo. Nulla fermentum sed augue a venenatis. Aliquam erat volutpat
vitae mollis nibh
quisque at blandit justo. Nulla fermentum sed augue a venenatis. Aliquam erat volutpat
vitae mollis nibh
quisque at blandit justo. Nulla fermentum sed augue a venenatis. Aliquam erat volutpat
Polimery
Plastica Sp. z o.o. wspiera akcję społeczną POLIMERY PRZYJAZNE CZŁOWIEKOWI, POTRZEBNE LUDZIOM
Niewiele jest spraw, co do których Polacy są zgodni. Jedną z nich jest fatalny publiczny wizerunek tworzyw sztucznych. Przemysł tworzyw stał się dyżurnym „chłopcem do bicia” w Polsce i UE, jest obwiniany za wszelkie błędy i wypaczenia naszej cywilizacji. Czas na podjęcie działań, które w rozsądnej perspektywie pozwolą zmienić negatywne społeczne nastawienie wobec tworzyw.
Przede wszystkim najwyższy czas na wyrugowanie z przestrzeni publicznej koszmarka językowego „tworzywa sztuczne”. Ten termin natychmiast odrzuca każdego normalnego człowieka, jego ocieplenie i zmiana wizerunku na pozytywny wydaje się niemożliwa, albowiem nic co „sztuczne” nie może być przyjazne człowiekowi. Dlatego proponujemy zastąpienie koszmarka językowego terminem „tworzywa polimerowe”, a szerzej POLIMERY. Nazwa POLIMERY oddaje istotę rzeczy i nie jest obciążona negatywnymi asocjacjami i skojarzeniami wytworzonymi wokół koszmarka językowego. Jest nawet sympatyczna i, jak widać po logo akcji, da się za jej pomocą komunikować treści mające odzwierciedlenie w rzeczywistości.
Pamiętajmy, że jak mówi Pismo, na początku było Słowo. Język ma kluczowe znaczeniu w międzyludzkiej komunikacji i tworzeniu korzystnego lub negatywnego wizerunku. Krótko mówiąc: język tworzy rzeczywistość.
Celem akcji jest rozpropagowanie terminu POLIMERY, który w niedalekiej przyszłości powinien zastąpić koszmarek językowy. Przemysł polimerowy musi się zacząć w końcu bronić. Polimery są niezbywalnym elementem naszej cywilizacji i tylko od człowieka, od nas samych zależy, jak będą nam służyć.
Wprowadzenie terminu POLIMERY do dyskursu publicznego pozwoli rozpocząć wiele pozytywnych działań wizerunkowych we współpracy z mediami i organizacjami społecznymi.
Zmiana wizerunku branży powinna ożywczo wpłynąć na sam biznes. Zejście z linii strzału ekologów, mediów, polityków, innych grup opiniotwórczych, powinno przynieść mierzalne biznesowe efekty. Taki stan rzeczy na pewno zostanie zauważony przez inwestorów zagranicznych. Wreszcie, może to być sygnał do konsolidacji przemysłu polimerowego, co zaowocuje wzrostem siły i znaczenia tej branży w całej gospodarce.






